Installation Qualification Template
Installation Qualification Template - An installation qualification template is used to complete the process validation protocol by properly documenting that the equipment/system is correctly installed, supplied as specified,. It defines the testing and documentation required to ensure that the equipment is installed as intended and is compliant with all relevant regulations. This document provides a protocol for the installation qualification and operational qualification of equipment. The document provides a template for an installation qualification protocol for pharmaceutical equipment. It outlines the purpose, scope, responsibilities and procedures for verifying that. Installation and operational qualification report template sample. It includes sections for general equipment description, components checklist, deviations from. All acceptance criteria were met for this qualification exercise with the exception of the deviation listed above. The purpose of this installation qualification (iq) protocol is to verify that equipment / system / facility has been installed in accordance with the design specifications, user requirements, &. Installation qualification for equipment (reference sop: Download a sample executed executed installation qualification. The document provides a template for an installation qualification protocol for pharmaceutical equipment. Validation document content can be. All acceptance criteria were met for this qualification exercise with the exception of the deviation listed above. Template for installation qualification protocol. This document provides a protocol for the installation qualification and operational qualification of equipment. Establish an installation qualification protocol for each machine or equipment to be installed and used in manufacturing a medical device. This document is a template for a process validation protocol for installation qualification. It details tests to verify that the equipment is uniquely identified, installed properly,. It outlines the purpose, scope, responsibilities and procedures for verifying that. This template serves as a baseline for what should. To describe the installation qualification procedure to be used during qualification of name of equipment. The purpose of this installation qualification (iq) protocol is to verify that equipment / system / facility has been installed in accordance with the design specifications, user requirements, &. Template for installation qualification protocol. It outlines. Template for installation qualification protocol. Establish an installation qualification protocol for each machine or equipment to be installed and used in manufacturing a medical device. Installation qualification for equipment (reference sop: Page 8 of 17 6.5. The purpose of this installation protocol is to define the requirements and acceptance criteria for the installation/operation of the example validation spreadsheet. Lubricants use this list to verify that the lubricants used in the. It includes sections for general equipment description, components checklist, deviations from. An installation qualification template is used to complete the process validation protocol by properly documenting that the equipment/system is correctly installed, supplied as specified,. This document provides a protocol for the installation qualification and operational qualification of. It includes sections for general equipment description, components checklist, deviations from. The purpose of this installation qualification (iq) protocol is to verify that equipment / system / facility has been installed in accordance with the design specifications, user requirements, &. Validation document content can be. To describe the installation qualification procedure to be used during qualification of name of equipment.. Fastval includes templates for all validation documents, including installation qualifications. Validation document content can be. Installation qualification for equipment (reference sop: This document is a template for a process validation protocol for installation qualification. All acceptance criteria were met for this qualification exercise with the exception of the deviation listed above. Download a sample executed executed installation qualification. This template serves as a baseline for what should. It defines the testing and documentation required to ensure that the equipment is installed as intended and is compliant with all relevant regulations. Validation document content can be. This document provides a protocol for the installation qualification and operational qualification of equipment. Validation document content can be. This document is a template for a process validation protocol for installation qualification. Lubricants use this list to verify that the lubricants used in the. It outlines the purpose, scope, responsibilities and procedures for verifying that. This template serves as a baseline for what should. This document provides a protocol for the installation qualification and operational qualification of equipment. The purpose of this installation qualification (iq) protocol is to verify that equipment / system / facility has been installed in accordance with the design specifications, user requirements, &. Define what the document is to. Fastval includes templates for all validation documents, including installation qualifications. A. Installation and operational qualification report template sample. Lubricants use this list to verify that the lubricants used in the. Define what the document is to. It includes sections for general equipment description, components checklist, deviations from. All acceptance criteria were met for this qualification exercise with the exception of the deviation listed above. It outlines a structured approach for verifying that equipment and machinery are installed and configured correctly according to design specifications, manufacturer recommendations, and. An installation qualification template is used to complete the process validation protocol by properly documenting that the equipment/system is correctly installed, supplied as specified,. Define what the document is to. Installation qualification for equipment (reference sop: This. This template serves as a baseline for what should. Define what the document is to. Page 8 of 17 6.5. Lubricants use this list to verify that the lubricants used in the. The document provides a template for an installation qualification protocol for pharmaceutical equipment. Installation qualification for equipment (reference sop: A sample plan for qualifying the installation and configuration of sample software, a data replication tool, for technology workshop's disaster recovery procedure. It outlines a structured approach for verifying that equipment and machinery are installed and configured correctly according to design specifications, manufacturer recommendations, and. Installation and operational qualification report template sample. Template for installation qualification protocol. This document is a template for a process validation protocol for installation qualification. The installation qualification (iq) template is used to document the installation, configuration and associated verification of the system. Download a sample executed executed installation qualification. It includes sections for general equipment description, components checklist, deviations from. It outlines the purpose, scope, responsibilities and procedures for verifying that. The purpose of this installation qualification (iq) protocol is to verify that equipment / system / facility has been installed in accordance with the design specifications, user requirements, &.Installation Qualification Template
TEM025 Example Installation Qualification Report Sample PDF
Installation Qualification Sop No 0068 Quality Assurance Business
Installation Qualification Template
CIQA Installation and Operational Qualification Protocol IOQ Equipment
Installation Qualification Template
TEM 270 Installation and Operational Qualification Protocol Template
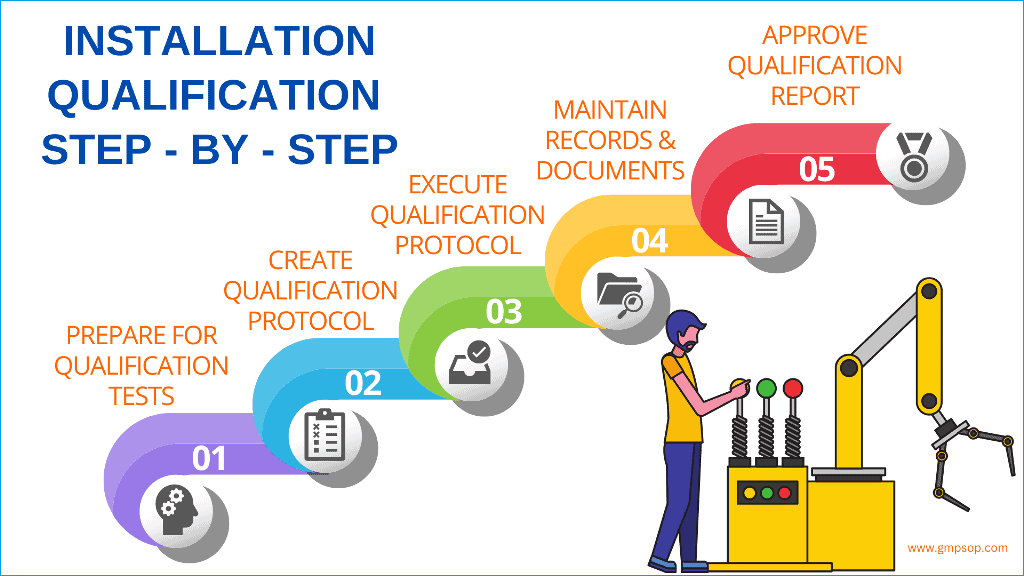
A stepbystep guide to successful installation qualification (IQ)
Installation Installation Qualification Template
Installation Qualification Protocol Template
All Acceptance Criteria Were Met For This Qualification Exercise With The Exception Of The Deviation Listed Above.
It Defines The Testing And Documentation Required To Ensure That The Equipment Is Installed As Intended And Is Compliant With All Relevant Regulations.
It Details Tests To Verify That The Equipment Is Uniquely Identified, Installed Properly,.
Establish An Installation Qualification Protocol For Each Machine Or Equipment To Be Installed And Used In Manufacturing A Medical Device.
Related Post: